
CMS Posts FY 2027 Hospice Wage Index Proposed Rule
CMS Posts FY 2027 Hospice Wage Index Proposed Rule
The FY 2027 Hospice Wage Index and Payment Rate Update and Hospice Quality Reporting Program Requirements [CMS-1851-P] was posted on the Federal Register Public Inspection desk on 4/2/2026. The rule was posted in the Federal Register on 4/6/2025. CMS will accept comments related to the proposed rule through June 1, 2026, 11:59 pm. Information about submitting comments appears at the beginning of the rule.
Providers are strongly encouraged to review the entire content of the proposed rule as not all detailed information is included in this summary. Here are the highlights of the proposed rule.
Payment update information:
- The FY 2026 proposed hospice payment update percentage is 2.4%.
- This proposed rule includes the proposed updates to the hospice wage index and makes the application of the updated wage data budget neutral for all four levels of hospice care.
- CMS also proposes that if more recent data becomes available after the publication of this proposed rule and before the publication of the final rule (for example, a more recent estimate of the inpatient hospital market basket percentage increase or productivity adjustment), CMS would use such data, if appropriate, to determine the hospice payment update percentage in the FY 2027 final rule.
- The proposed hospice cap amount for the FY 2027 cap year is $36,210.11.
- CMS also proposes that if more recent data becomes available after the publication of this proposed rule and before the publication of the final rule (for example, a more recent estimate of the hospice payment update percentage), CMS would use such data, if appropriate, to determine the hospice cap amount in the FY 2027 final rule.
Proposed FY 2027 Hospice RHC Payment Rates
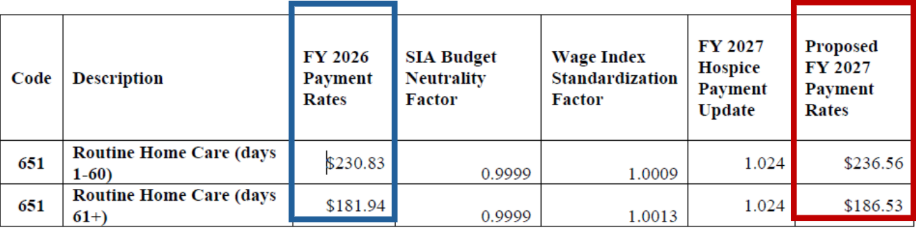
Proposed FY 2027 Hospice CHC, IRC, and GIP Payment Rates
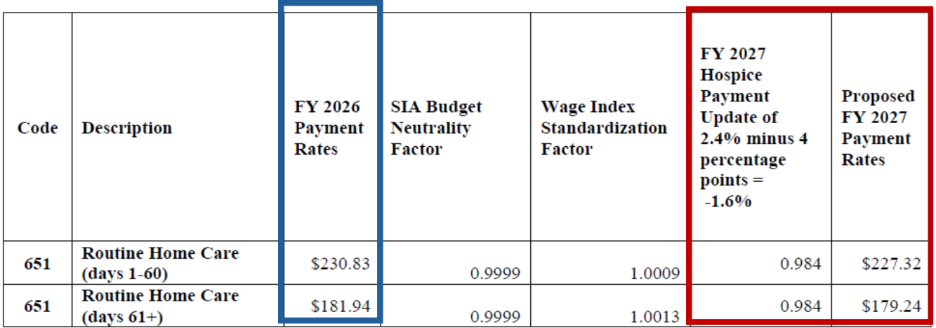
Proposed FY 2027 Hospice RHC Payment Rates for Hospices That DO NOT Submit the Required Quality Data (the proposed FY 2027 rates for hospices that do not submit the required quality data would be updated by -1.6 percent, which is the proposed FY 2027 hospice payment update percentage of 2.4 percent minus 4 percentage points)
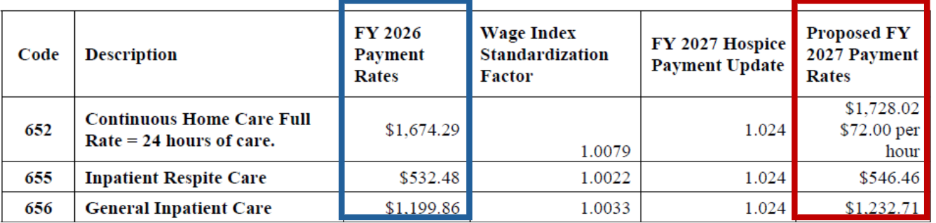
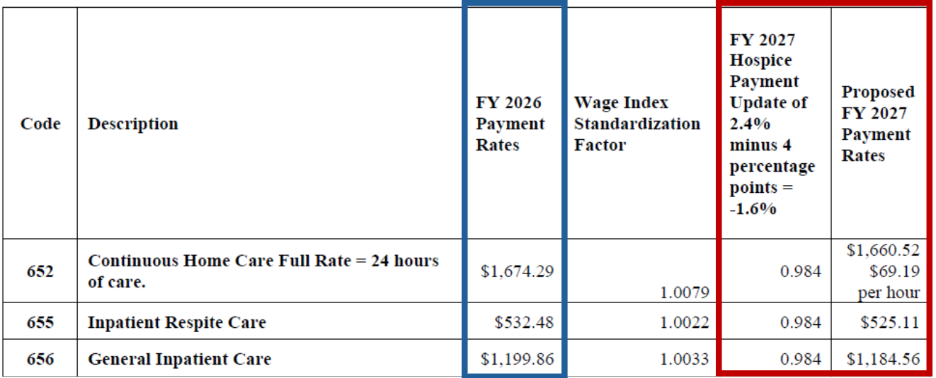
Proposed FY 2027 Hospice CHC, IRC, and GIP Payment Rates for Hospices That DO NOT Submit the Required Quality Data (the proposed FY 2027 rates for hospices that do not submit the required quality data would be updated by -1.6 percent, which is the proposed FY 2027 hospice payment update percentage of 2.4 percent minus 4 percentage points)
Medicare Non-Hospice Spending
- Background – Medicare Parts A & B
- As referenced in federal hospice regulations at § 418.64, a hospice must routinely provide all core services directly by hospice employees and they must be provided in a manner consistent with acceptable standards of practice. Under the current payment system, hospices are paid for each day that a beneficiary is enrolled in hospice care, regardless of whether services are rendered on any given day. Additionally, when a beneficiary elects the Medicare hospice benefit, he or she waives the right to Medicare payment for services related to the treatment of the terminal illness and related conditions, except for services provided by the designated hospice and the attending physician.
- The Medicare hospice benefit is designed so beneficiaries rarely need services or medications outside of what hospice provides. Since 1983, it has been emphasized that nearly all necessary care for terminally ill patients should come from hospice (referenced in multiple Federal Registers). Hospices must inform patients that outside services are rare and unusual, as detailed in § 418.24(b)(3).
- Since the implementation of the hospice election statement addendum requirement in FY 2020 (84 FR 38484), which must be provided upon request, Medicare non-hospice spending for beneficiaries who have elected the hospice benefit has shown substantial and consistent growth. Specifically, Medicare paid over $2.8 billion in non-hospice spending during a hospice election in FY 2024 for items and services under Parts A, B, and D.
- Medicare payments for non-hospice Part A and Part B items and services received by hospice beneficiaries during a hospice election increased from nearly $790 million in FY 2020 to over $2 billion in FY 2024 (see Figure B1). This represents an increase in non-hospice Medicare spending for Parts A and B of nearly $1.3 billion, or 160 percent. The most substantial increase in a single year occurred from FY 2023 to FY 2024, which demonstrated an increase in non-hospice Medicare spending for Part A and Part B items and services of $770 million, or 60 percent.
- In FY 2024, the total beneficiary cost sharing amount for beneficiaries electing the hospice benefit was $510 million for Parts A and B. In FY 2024, beneficiaries receiving hospice services from for-profit hospices had, on average, nearly 167 percent higher non-hospice spending per day compared to beneficiaries under non-profit hospice care.
- This represents a significant increase from FY 2022, when beneficiaries receiving hospice services from for-profit hospices had, on average, 60 percent higher non-hospice spending per day compared to beneficiaries under non-profit hospice care.
- The most dramatic increase in billing related to carrier/physician supply. From FY 2020 to FY 2024, nonhospice spending related to carrier/physician supply increased 317.5 percent with a notable single year spike from FY 2022 to FY 2023 of 63.5 percent, and the largest increase in one year occurred from FY 2023 to FY 2024 with an increase of 90.8 percent. The diagnosis code for carrier claims with the largest increase in spending in FY 2024 was for pressure ulcers, largely associated with skin substitutes, which accounted for 47 percent, almost half of the carrier claim spending.
- CMS found notable consistent increases in outpatient and inpatient services in recent years. Non-hospice spending related to outpatient services increased 40.4 percent and inpatient services increased by 26.9 percent in the same time frame. Additionally, they found that 30.1 percent and 25.9 percent of the non-hospice spending that occurred in FY 2024 was related to the primary hospice diagnosis of Alzheimer’s disease/dementia/Parkinson’s and heart conditions (Congestive Heart Failure and other heart disease.
- Background – Medicare Part D
Hospices are responsible for covering drugs and biologicals related to the palliation and management of the terminal illness and related conditions while the patient is under hospice care.
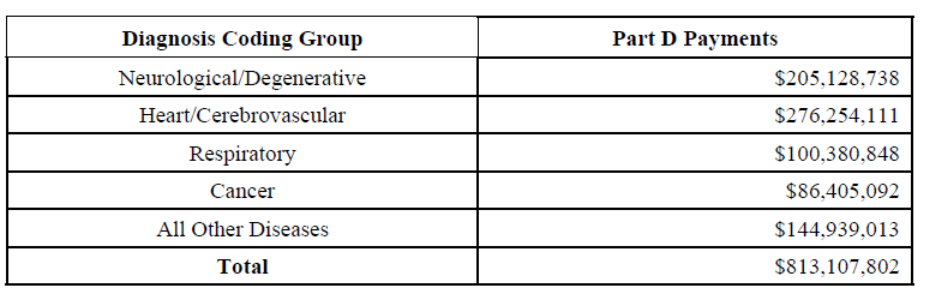
- Non-hospice spending for Part D drugs increased from $552.9 million in FY 2020 to $813.1 million in FY 2024, which represents an increase of over a 47 percent.
- The portion of the FY 2020 to FY 2024 Part D spending that was paid by Medicare is the sum of the Low-Income Cost-Sharing Subsidy and the Covered Drug Plan Paid Amount, approximately $3.3 billion. The beneficiary cost sharing amount was approximately $335.1 million.
- CMS also noted hospice beneficiaries with principal diagnoses of neurological and degenerative diseases, circulatory and cerebrovascular diseases, respiratory diseases, and neoplasms have received clinically indicated services for these conditions outside the hospice benefit.
- CMS analyzed the same principal diagnosis coding groups for Part D drugs paid outside of the hospice benefit (see data chart below for spending amounts).
Medicare Payments for Non-Hospice for Part D During Hospice Elections Provided to Hospice Beneficiaries (Part D Claims), FY2024
- Service and Spending Variation Index (SSVI)
- CMS currently monitors and publicly shares data related to hospice utilization. Using the most recent, complete claims data, CMS analyzes Medicare spending, utilization by level of care, lengths of stay, live discharge rates, and skilled visits during the last days of life.
- By analyzing hospice utilization and other metrics, CMS can evaluate the behaviors of hospices to combat potential risks to the integrity of the Medicare program.
- Analyzing these particular Medicare hospice metrics together is important because patterns across them can signal potential program integrity risks, inappropriate utilization, or quality of care concerns, especially when they deviate substantially between different hospices or from expected norms.
- For example, long lengths of stay combined with high live discharge rates may signal inappropriate enrollment of ineligible beneficiaries.
- Low number of visits, shorter visits, or fewer weekend visits may indicate minimal service provision.
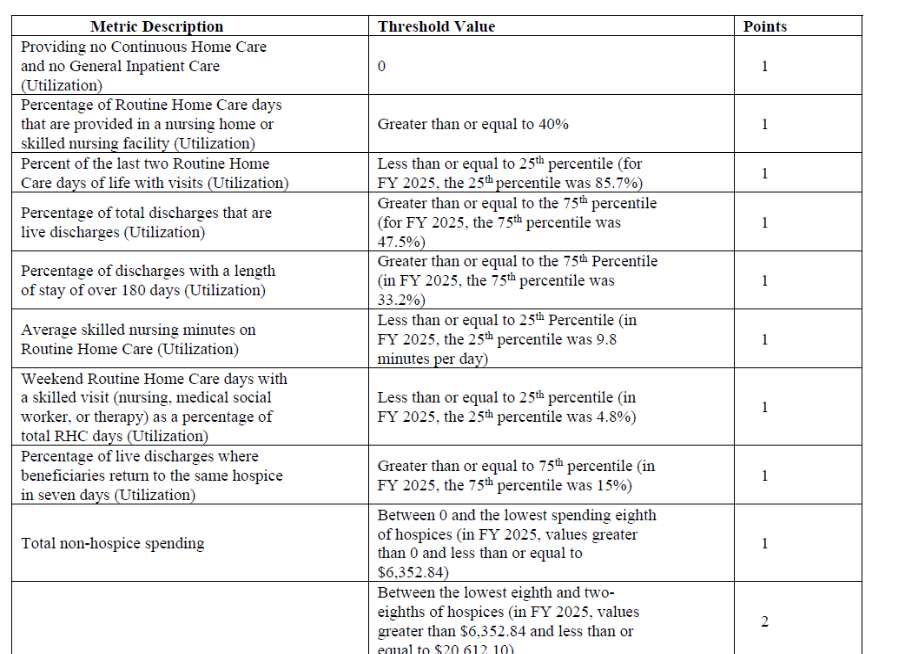
- CMS developed a scoring system, the Service and Spending Variation Index (SSVI), that is calculated using nine claims-based measures, each representing different aspects of hospice utilization as well as non-hospice spending.
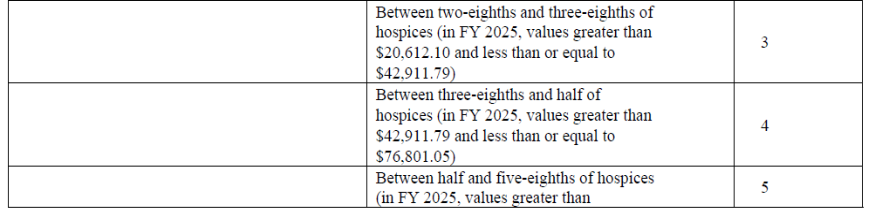
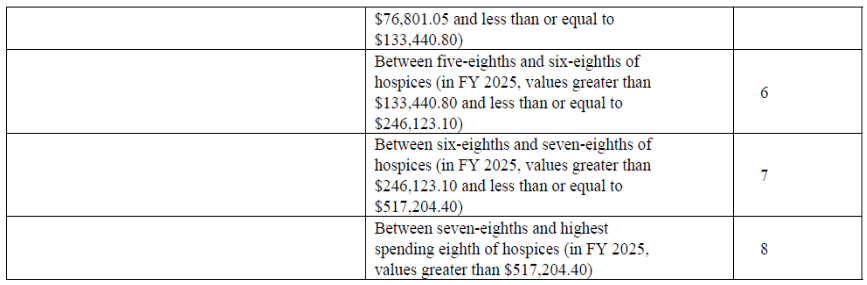
- To calculate the SSVI score, CMS first determined a threshold for each of the nine metrics (See Table below). For the non-hospice spending component of the SSVI score, CMS created eight separate thresholds for total non-hospice spending, as the degree to which a hospice spends outside of the hospice benefit can indicate varying levels of concern.
- For example, a hospice with higher non-hospice spending levels receives a higher number of points than a hospice with about 12.5 percent less non-hospice spending.
- Metrics related to utilization reflect visit and discharge patterns.
- The SSVI can be used to identify hospices that are outliers across many different utilization metrics and those that have a high level of non-hospice spending.
- CMS established thresholds using percentiles. For most of the individual measures, they established the threshold at the top or bottom 25 percent of the distribution.
- The objective of the SSVI is not to evaluate hospices based on a single metric, but to identify hospices that are outliers across multiple independent metrics.
- The lowest SSVI score a hospice can receive is zero, that is, a score of zero for each of the nine metrics, and the maximum SSVI score is 16, that is, with the highest points assigned for each of the nine metrics. A higher SSVI score represents a potential higher level of concern, as this may signal potential program integrity risks or inappropriate utilization, especially when a hospice’s SSVI score is substantially higher than its peers.
Table: Description of SSVI Metrics, Threshold Values, and Points Allocated
Publication and Update of SSVI Scores
- CMS is publishing the Service and Spending Variation Index (SSVI) scores calculated from data for fiscal years (FYs) 2024 and 2025. These years were selected because they represent the most recent and complete sets of claims data available.
- The FY 2024 hospice SSVI includes 6,409,155 hospice claims, representing 6,735 hospices and a total of 148,012,785 hospice days. The FY 2025 hospice SSVI includes 6,750,840 hospice claims, representing 6,642 hospices and a total of 156,514,386 hospice days.
- CMS may update the FY 2024 and FY 2025 SSVI scores with any revisions deemed appropriate based on comments received regarding this proposed rule. Any updates would be published with the FY 2027 Hospice Wage Index and Rate Update final rule. In subsequent rulemaking cycles, CMS will continue to publish updated SSVI scores, using the most recent claims data, along with the final rule.
Table: The distribution of the number of hospices by their total SSVI score for hospices in FYs 2024 and 2025 claims
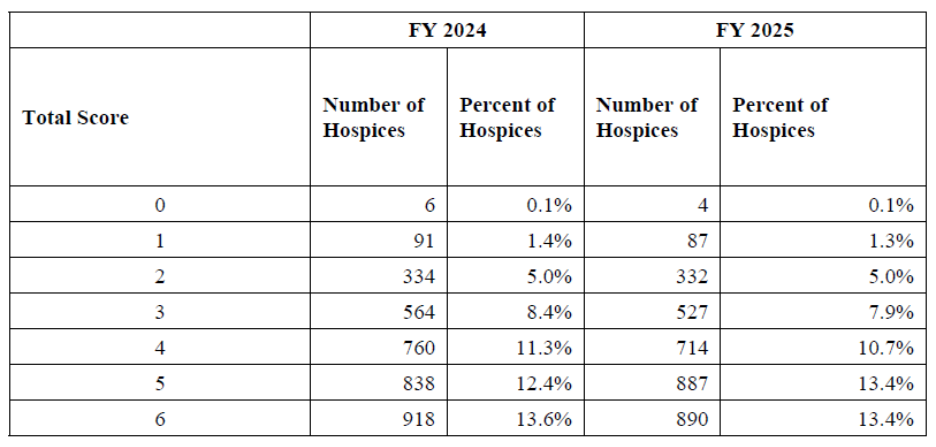
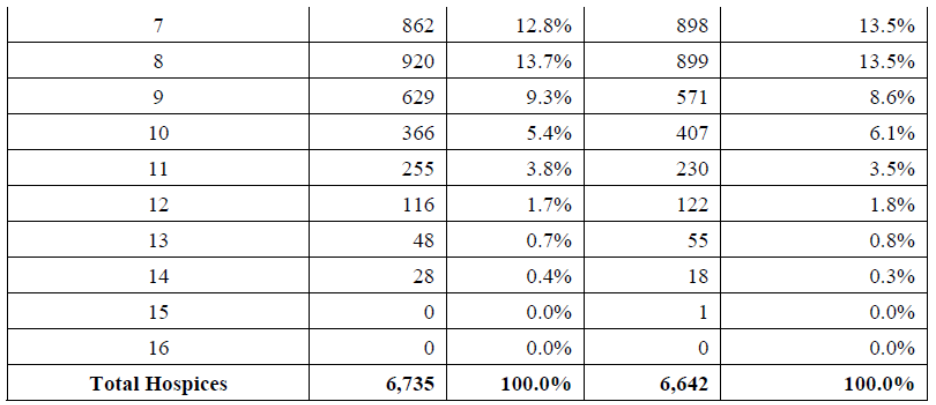
- CMS will publish metrics, SSVI scores for FYs 2024 and 2025, claims-based data, and methodology documentation on the Hospice Center webpage at https://www.cms.gov/medicare/enrollment-renewal/providerssuppliers/hospice-center
- The aim is to identify hospice vulnerabilities to guide program integrity actions such as medical reviews, education, investigations, and possible administrative actions like payment suspension or revocation for fraudulent hospices.
- The public will benefit from greater transparency, helping beneficiaries and families make informed end-of-life care decisions.
Opportunity for Feedback to CMS: Feedback is welcomed on SSVI metrics, threshold values, and point assignments.
Proposed Election Statement Addendum Changes
- In the FY 2020 Hospice Wage Index and Payment Rate Update and Hospice Quality Reporting Requirements final rule (84 FR 38484), CMS finalized a policy, for elections beginning on and after October 1, 2020, that requires hospices to provide a hospice election statement addendum to beneficiaries, their representatives, non-hospice providers, or Medicare contractors, upon request.
- The purpose of the addendum is to notify the hospice beneficiary (or representative) of those conditions, items, services, and drugs the hospice will not be covering because the hospice has determined they are unrelated to the beneficiary’s terminal illness and related conditions.
- A model hospice election statement addendum is available on the Hospice Center webpage at https://www.cms.gov/Center/Provider-Type/Hospice-Center.
Proposed change to regulation:
CMS proposes to require that hospices provide the hospice election statement addendum to all Medicare beneficiaries at the time of hospice election for hospice elections beginning on or after October 1, 2026.
Rationale for regulatory change – significant increases in non-hospice spending patterns suggest that the current framework, where the hospice election statement addendum is provided only upon request, has not achieved the intended accountability objective of ensuring that hospices provide virtually all care needed by terminally ill individuals as required under the comprehensive and holistic Medicare hospice benefit.
Proposed § 418.24 Election of hospice care.
(b)(6) For Hospice elections beginning on or after October 1, 2026, the hospice must provide the individual (or representative) an election statement addendum, as set forth in paragraphs (c) and (d) of this section, which includes any conditions, items, services, and drugs the hospice has determined to be unrelated to the individual’s terminal illness and related conditions and would not be covered by the hospice.
(c) Content of hospice election statement addendum. For hospice elections beginning on or after October 1, 2026, the hospice must provide the individual (or representative) an election statement addendum. The election statement addendum (and its updates) must include the following:
(9) Name and signature of the individual (or representative) and date signed, along with a statement that signing this addendum (and its updates) is only acknowledgement of receipt of the addendum and not the individual’s (or representative’s) agreement with the hospice’s determinations. If the individual (or representative) refuses to sign the addendum, the hospice must document on the addendum the reason the addendum was not signed and the addendum would become part of the patient’s medical record. The addendum must also be available for non-hospice providers and Medicare contractors, although non-hospice providers and Medicare contractors are not required to sign the addendum.
(10) Date the hospice furnished the addendum to the individual (or representative).
(d) Timeframes for the hospice election statement addendum. (1) For hospice elections beginning on or after October 1, 2026, the hospice must provide the individual (or representative) an election statement addendum, in writing, as set forth in paragraph (c) of this section, at the time of the hospice election (that is, within the first 5 days of the effective date of the hospice election). The hospice must also file this information with the election statement, as set forth in paragraphs (a) and (b) of this section, to be available for the individual (or representative), non-hospice providers, and Medicare contractors.
(2) If there are any changes to the plan of care during the course of hospice care that impact the addendum determinations, the hospice must update the addendum, within 3 days, with the contents described in paragraph (c) of this section, and provide these updates, in writing, to the individual (or representative), as well as update the addendum on file in order to communicate these changes to the individual (or representative), non-hospice providers, and Medicare contractors.
(3) If the individual dies, revokes, or is discharged within the required timeframe for providing the addendum (and its updates) (as outlined in paragraphs (d)(1) and (2) of this section), and before the hospice has provided the addendum (and its updates), the addendum would not be required to be provided, in writing, to the individual (or representative). The hospice must note the reason the addendum (and its updates) was not completed and/or provided, in writing, to the individual (or representative) and this note would become part of the patient’s medical record. If completed, the hospice must still file the addendum (and its updates) with the election statement, as set forth in paragraphs (a) and (b) of this section, to be available for the individual (or representative), non-hospice providers, and Medicare contractors.
(4) If the individual dies, revokes, or is discharged prior to signing the addendum (or its updates) (as outlined in paragraphs (d)(1) and (2) with the required contents described in paragraph (c) of this section), the addendum would not be required to be signed in order for the hospice to receive payment. The hospice must note (on the addendum itself) the reason the addendum (and any updates) was not signed and the addendum would become part of the patient’s medical record.
Opportunity for feedback: CMS solicits comments on this proposal.
Proposed Clarifying Regulation Text Changes
- Discharge from Hospice Care
Proposed change to the regulation: CMS proposes additions to § 418.26(b) to state that the hospice may also obtain the written physician’s discharge order from the physician designee, as defined at § 418.3, or physician member of IDG.
Proposed § 418.26 Discharge from hospice care.
* * * * *
(b) Discharge order. Prior to discharging a patient for any reason listed in paragraph (a) of this section, the hospice must obtain a written physician’s discharge order from the hospice medical director (or physician designee, as defined at § 418.3) or physician member of the interdisciplinary group. If a patient has an attending physician involved in his or her care, this physician should be consulted before discharge and his or her review and decision included in the discharge note.
Opportunity for feedback: CMS requests comments on the proposed additions to § 418.26(b).
- Face-to-Face Encounter
- Section 6209(f)(1)(A) of the CAA – Amends the regulatory allowance to use telehealth by a hospice physician or hospice nurse practitioner to conduct a face-to-face encounter for the sole purpose of recertifying the patient’s eligibility for hospice has been extended through December 31, 2027.
- Section 6209(f)(1)(B) of the CAA prohibits telehealth for face-to-face encounters in certain hospice cases starting January 31, 2026, including individuals in areas under hospice enrollment moratoriums, those cared for by providers under enhanced oversight, or encounters by unenrolled or non-opt-out hospice physicians or nurse practitioners.
- Section 6209(f)(2) requires that from January 1, 2027, hospice claims for telehealth encounters must include modifiers or codes specified by the Secretary to show the encounter was via telehealth.
Proposed change to regulation:
CMS proposes to amend § 418.22(a)(4)(ii) to require the hospice to collect data reflecting face-to-face encounters furnished using telecommunications technology, which includes, at a minimum, the use of audio and video equipment permitting two-way, real-time interactive communication between the patient and the distant site hospice physician or hospice nurse practitioner, and the hospice would do so by reporting a G-code identifying that a face-to-face encounter was furnished using such technology, that is, telehealth.
Proposed § 418.22 Certification of terminal illness.
(a) * * *
(4) * * *
(ii) During a Public Health Emergency, as defined in §400.200 of this chapter, or through December 31, 2027, whichever is later, if the face-to-face encounter conducted by a hospice physician or hospice nurse practitioner is for the sole purpose of hospice recertification, such encounter may occur via telecommunications technology and is considered an administrative expense. Telecommunications technology means the use of interactive multimedia communications equipment that includes, at a minimum, the use of audio and video equipment permitting two-way, real-time interactive communication between the patient and the distant site hospice physician or hospice nurse practitioner. For face-to-face encounters occurring on or after January 1, 2027, hospices must report any such encounters occurring via telecommunications technology on the claim, in accordance with guidance issued by CMS. Beginning January 31, 2026, telehealth may not be used for the face-to-face recertification encounter if any of the following conditions apply:
(A) The hospice patient is located in an area subject to a hospice enrollment moratorium under section 1866(j)(7) of the Act;
(B) The patient is receiving care from a hospice provider that is subject to enhanced oversight pursuant to section 1866(j)(3) of the Act; or
(C) The face-to-face encounter is conducted by a hospice physician or nurse practitioner who is not enrolled in Medicare under section 1866(j) and is not an opt-out physician or practitioner (as defined in section 1802(b)(6)(D) of the Act.
* * * * *
3. Section 418.24 is amended by revising paragraphs (b)(6), (c) introductory text, (c)(9)
Opportunity for feedback: CMS solicits comments on these proposed amendments and on the use of the new G-code identifying face-to-face encounters furnished via telehealth.
Requests for Information (RFI) on Medicare Services and Payment Structure
- Request for Information on Ways to Enhance the Provision of Palliative Care Outside of Hospice Care: Current Coverage, Billing Practices, and Opportunities for Improvement
Interested party feedback is essential for guiding CMS toward policies that expand access to high-quality community palliative care without requiring legislative reform or the creation of an entirely new benefit.
- Medicare defines palliative care as patient and family centered care that optimizes quality of life by anticipating, preventing, and treating suffering. Palliative care throughout the continuum of illness involves addressing physical, intellectual, emotional, social, and spiritual needs and to facilitate patient autonomy, access to information, and choice (§ 418.3).
- CMS states that although Medicare does not currently offer a dedicated palliative care benefit, because palliative services are offered across existing Medicare programs, they are interested in soliciting public feedback regarding ways in which they can optimize current coverage and billing practices under various outpatient or home-based benefits to result in more cohesive, integrated, person-centered care as beneficiaries approach hospice care.
- CMS wants to understand how Medicare providers currently support palliative care, how providers bill for these services, and where gaps persist is critical to strengthening community-based palliative care within today’s regulatory and payment structure.
- Understanding Billing Practices and Delivering Palliative Care
- CMS is interested in understanding how community providers bill for palliative services, which CPT or HCPCS codes they rely on, and what barriers they face in using ACP, care management, or telehealth codes.
- There are multiple specific questions related to this RFI in this section of the rule and providers should respond to each one in their comments.
- Understanding Program and Beneficiary Needs
- In addition to providing feedback on billing practices, interested parties can offer insight into broader systemic challenges, staffing limitations, claim denials, and palliative services they provide but cannot bill for under Medicare’s current structure.
- There are multiple specific questions related to this RFI in this section of the rule and providers should respond to each one in their comments
Opportunity for feedback: In addition to the questions in each section of the RFI, CMS solicits input on any additional targeted enhancements within current benefits, such as expanding billable services, simplifying documentation, standardizing definitions, or increasing beneficiary education that could meaningfully expand access to palliative care services.
- Request for Information Regarding Construction of a Hospice Specific Wage Index (please review this section carefully as this is only a high level summary)
The hospice wage index is used to adjust payment rates for hospices under the Medicare program to reflect local differences in area wage levels, based on the location where services are furnished, as determined by the Secretary, in accordance with sections 1814(i)(1)(A) and 1814(i)(2)(D) of The Act.
- CMS received numerous comments regarding the use of the Inpatient Prospective Payment System (IPPS) wage index to adjust for the geographic variation of wages for hospice staff through the annual hospice rulemaking. Specifically, commenters have stated that the IPPS wage index uses data from four FYs prior to the current payment year and that the time lag may underestimate the changes in relative wages for hospice staff.
- CMS received recommendations from MedPAC to include all-employer, occupation-level wage data to establish different weights for setting-specific occupational labor mix to capture labor costs faced by all employers of the related occupations.
Opportunity for feedback: looking for feedback on how the BLS OEWS data, and other public data can be used to construct a hospice specific wage index. CMS requests input to understand the advantages and limitations of the suggested approach in using BLS data and cost reports to support the construction of a hospice specific wage index.
- Request for Information Regarding Medical Aid in Dying (MAID)
- The Assisted Suicide Funding Restriction Act of 1997 (Pub. L. 105-12, April 30,1997) prohibits the use of Federal funds (through Medicare, Medicaid, and other Federal programs) to provide or pay for any health care item or service, or health benefit coverage, for the purpose of causing, or assisting to cause, the death of any individual including mercy killing, euthanasia, or assisted suicide, sometimes referred to as “medical aid in dying” (MAID).
- MAID is not legal under Federal law; however, it is considered an end-of-life option for terminally ill adults to self-administer life-ending medication prescribed by a physician in certain states where it is allowed under State law
Opportunity for feedback: Because of State requirements (where MAID is allowed under State law) that a patient be terminally ill, CMS is interested in hearing from hospice providers and other interested parties about any issues that may arise when a Medicare hospice patient requests MAID. CMS asks 3 specific questions in this section. If you are commenting, address these questions. They also welcome any additional information regarding hospices’ experience with patients choosing to utilize MAID, with the expectation that hospice providers and staff are adhering to Federal law.
Updates to the Hospice Quality Reporting Program (HQRP)
- Updates Regarding the HOPE
- As finalized in the FY 2025 Hospice Wage Index final rule (89 FR 64202), public reporting of the HOPE quality measures would be implemented no earlier than FY 2028.
- CMS still expects to begin public reporting in November 2027, but this may change based on the quality and reportability of the data as determined by the CMS analysis of CY 2026 data, which would begin in CY 2027.
- HOPE assessment timeliness threshold:
- To meet the assessment timeliness threshold under the Annual Payment Update (APU), hospices must achieve a timely submission rate of 90 percent or higher for FY2027. This means that 90 percent of all HIS and/or HOPE assessments must be submitted to and accepted by CMS within 30 days of the patient’s admission or discharge date.
- Due to the newness of the HOPE assessment along with the migration to the iQIES platform, CMS has granted a waiver to all HOPE assessments dated October 1, 2025, through December 31, 2025, and as a result, all HOPE assessments with a target date in 2025 will be considered timely.
- Proposal to Add an Icon for Hospices on Medicare.gov Compare Tool to Indicate Failure to Meet Reporting Requirements
Despite the APU penalty increase from 2 percent to 4 percent in Fiscal Year (FY) 2024, CMS has not observed a significant improvement in the number of hospices meeting the QRP reporting requirements.
- To help provide additional information and context to consumers, while also serving to highlight non-compliant hospices, CMS is proposing to add an icon identifying hospice facilities, on the Medicare.gov Compare Tool, that have failed to meet reporting requirements for the HQRP.
- The proposed icon will identify hospices failing to submit any data or submitting less than the required 90 percent of HOPE submissions within 30 days of the patient’s admission or discharge date within a year period.
- The icon would provide an incentive for hospices to comply with the quality data submission requirements, while also communicating to consumers that CMS may not have enough data to adequately determine the quality of the hospice.
- CMS proposes to add the icon to the Medicare.gov Compare Tool no earlier than FY 2028 (October 1, 2027) to align with the addition of HOPE data to the Medicare.gov site, and the data will be based on CY 2026 APU submission data received from January 1, 2026, through December 31, 2026.
- The proposed icon will be added or removed on an annual basis to give hospices an ample amount of time to review and correct data, and to comply with the 90 percent threshold.
- The proposed icon would be visible both on the provider search page, as well as the individual hospice page on the Compare Tool, similar to how the icons appear for nursing homes and hospitals on the Medicare.gov site.
- Additional information will be added to the Compare Tool to ensure consumers are aware of what the icon means and how it should be taken into consideration.
- The aim of the icon would be to notify consumers that the hospice did not report sufficient data to CMS. Additional information about HQRP reporting requirements and APU penalty can be found on the HQRP Requirements and Best Practices website at https://www.cms.gov/medicare/quality/hospice/hqrp-requirements-and-best-practices
Opportunity for feedback: CMS invites public comment on our proposal to include an icon for hospices on the Medicare.gov Compare Tool to identify hospices that do not comply with the quality data submission requirements for the APU.
- Future Measures Update
A Technical Expert Panel (TEP) convened in November 2024 provided input on potential new or potential HCI indicators and based on that feedback. This report can be found at https://www.cms.gov/files/document/fall-2024-hqrp-tep-summary-report508c.pdf. Based on this feedback, along with input from other interested parties and additional analysis of the measure and its indicators, CMS is currently considering making changes to the HCI measure and plans to submit the updated measure to the 2026 MUC list. The aim of re-specifying the HCI measure is to make it more useful and important to providers and consumers.
- Form, Manner, and Timing of Quality Measure Data Submission
Submission requirements are codified at § 418.312.

- Submission of Data Requirements
Hospices must submit 90% of required HOPE records within 30 days of each event (admission, discharge, or up to two HUV timepoints based on patient stay) to support quality measures. Most noncompliance occurs due to missing this threshold. Numerous trainings are available online anytime to help hospice staff meet HQRP requirements. For updated resources, visit the HQRP website: https://www.cms.gov/medicare/quality/hospice, view training materials at HQRP Training and Education Library, or find more information at Requirements and Best Practices.
Collection of Information Requirements
CMS is also soliciting public comment on each of these issues for the following in this section of the proposed rule:
- Wage Data Used for the Proposed Mandatory Election Statement Addendum
- Proposed Information Collection Requirements (ICRs)
- Estimated Hospice Burden Related to Mandatory Election Statement Addendum
Read each section carefully for detailed information.
Response to Comments
Because of the large number of public comments, CMS normally receives on Federal Register documents, CMS is not able to acknowledge or respond to them individually. CMS will consider all comments received by the comment due dates, and, when they proceed with a subsequent document, they will respond to the comments in the preamble to that document.
View the Proposed Rule on the Federal Register’s Public Inspection Desk.
For further information, see the CMS summary of the hospice proposed rule.
Questions about the content of this rule? Contact CHAP